In vitro fertilization (IVF) has been the primary treatment for infertility since the birth of the first “test-tube baby” in 1978, helping millions of families realize their dream of parenthood. Over the past four decades, IVF has undergone remarkable innovations—from advances in ovarian stimulation protocols and embryo culture systems to intracytoplasmic sperm injection (ICSI) and vitrification cryopreservation. Despite these advancements, however, cumulative live birth rates have seen little substantial improvement.
Now, a groundbreaking study is changing that narrative. Researchers have developed a novel automated device capable of recovering additional oocytes from follicular fluid that would traditionally be discarded. In over half of all IVF cases, oocytes are lost during the process—and this team has already helped one mother give birth to a healthy baby using this revolutionary technology.

Understanding Microfluidic Technology
Microfluidics is a technology that precisely controls minute amounts of fluids through microscale channels—essentially building an intricate “miniature pipeline system” on a chip. While this technology has long been widely used in medical diagnostics and other fields, its application in reproductive medicine has faced persistent challenges: handling clinically complex samples that are both compositionally diverse and relatively large in volume.
The device developed in this study comprises four core functional modules. First, a filtration module blocks larger tissue debris present in follicular fluid. Second, a denudation module uses precisely designed microchannels to remove the cumulus cells surrounding oocytes. Third, a concentration module removes the majority of follicular fluid to increase oocyte density. Finally, a capture module collects intact oocytes at a stable flow rate. The entire process takes approximately 30 minutes, and the closed-system design ensures sterility throughout operation.
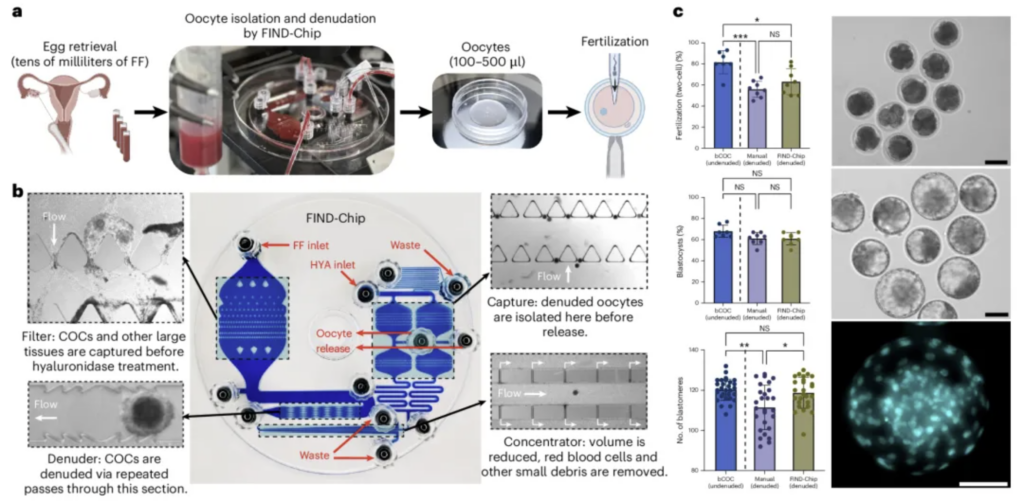
Study Results
The research team conducted a large-scale clinical study across four reproductive medicine centers, enrolling 582 patients undergoing IVF treatment. The patients had a median age of 36 years and a median clinical oocyte count of 12 oocytes.
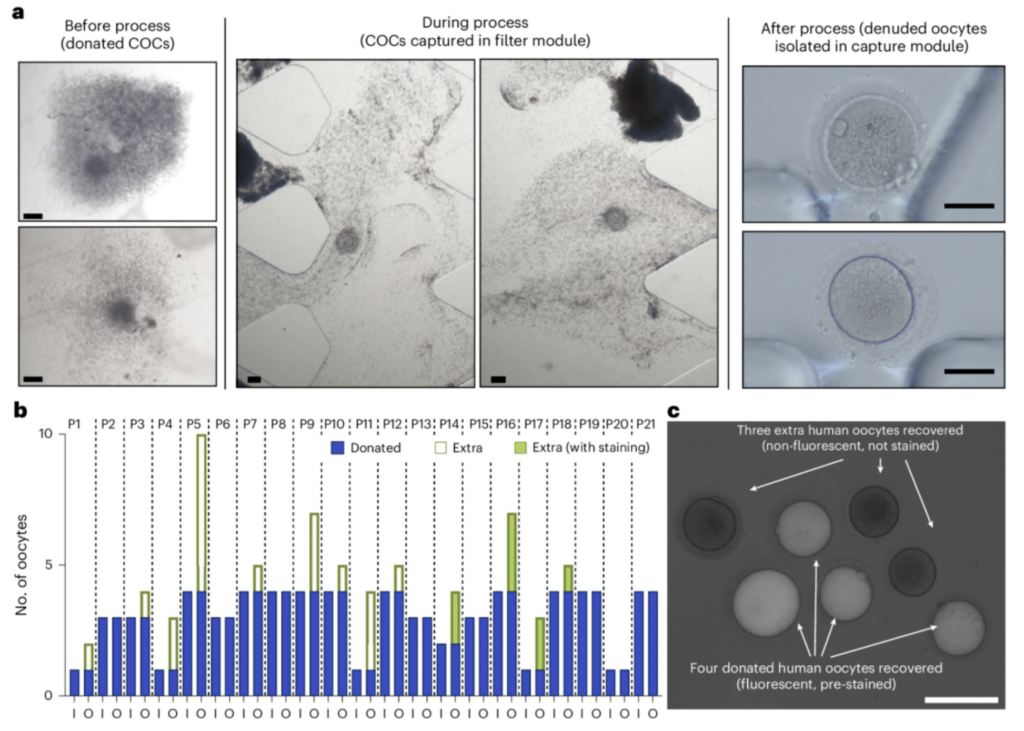
The results were striking. Additional oocytes were found in the follicular fluid of 54.3% of patients, yielding a total of 583 extra oocytes. Approximately 41% of these recovered oocytes were in mature condition and immediately ready for fertilization.
The study also evaluated the quality of these additional oocytes. The fertilization rate in the new device group reached 66.7%, comparable to the conventional manual selection group. Furthermore, the blastocyst development rate (early blastocyst and above) was 63%—significantly higher than the 49% observed in the conventional group.
The most milestone achievement: a healthy baby was born. During IVF treatment, this patient’s mother had an oocyte recovered from what would have been discarded follicular fluid using the new device, ultimately giving birth to a healthy infant.
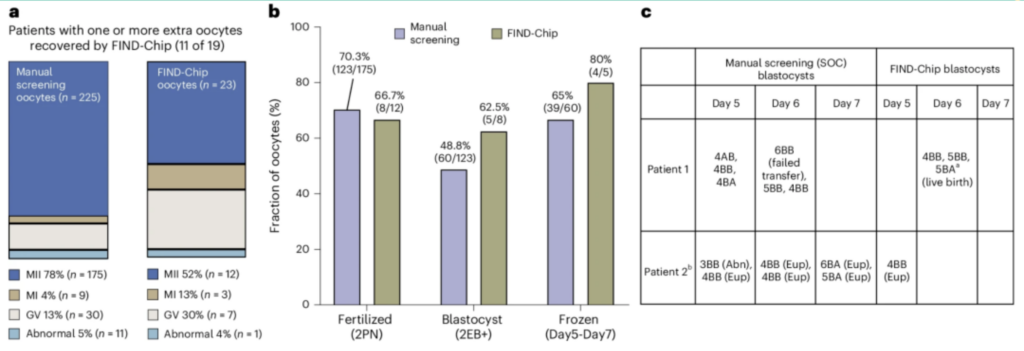
Clinical Significance
For families currently undergoing or considering IVF treatment, this technology means more opportunities and higher success rates.
For patients with low oocyte yields, even gaining just 1 to 2 additional oocytes can significantly improve cumulative live birth rates. Research shows that among patients with lower oocyte counts, approximately 35% to 51% have additional oocytes that can be recovered.
For patients with moderate oocyte yields, recovered oocytes provide more options for selecting high-quality embryos, increasing the success rate of single embryo transfers.
This technology will also transform reproductive medicine practice. Automation reduces reliance on highly skilled technicians, helping to narrow the performance gap between different clinics and driving IVF treatment toward greater standardization.
Limitations and Future Directions
As with any emerging technology, there are areas for improvement. Currently, the device releases denuded oocytes, requiring patients to undergo ICSI for fertilization—this somewhat limits treatment options. Additionally, oocytes are immediately denuded after retrieval, lacking the traditional “waiting period” of conventional protocols; the long-term implications of this require further study.
Despite these considerations, the technology shows tremendous promise. Future developments include further optimizing device design to improve sample processing completeness, conducting larger-scale multicenter clinical trials for more robust evidence, and extending this technology to resource-limited regions to benefit more patients.
Conclusion
This breakthrough offers fresh perspectives for the IVF field. It reveals a significant issue in current clinical practice—oocyte loss that could otherwise be prevented—and provides a viable solution. The data clearly shows that over half of IVF cases contain recoverable oocytes that are routinely missed, and these recovered oocytes demonstrate normal fertilization and developmental potential. The success of the live birth case proves the feasibility of this approach.
For countless families battling infertility, every technological advancement represents new hope. We look forward to more research validating this technology’s long-term effects, including blastocyst utilization rates, implantation rates, and cumulative live birth rates. Only through larger-scale, longer-term clinical validation can this technology truly mature and bring joy to more families.

