In the field of assisted reproductive technology (ART), sperm motility assessment represents a critical component of male fertility evaluation. Traditional computer-aided sperm analysis (CASA) systems track sperm movement trajectories from video footage and calculate kinematic parameters, providing a relatively objective assessment tool for clinical practice. However, the fundamental smoothing algorithms employed by these systems suffer from inherent limitations: they are prone to over-smoothing or under-smoothing, leading to errors in the calculation of key parameters such as beat cross frequency (BCF) and amplitude of lateral head displacement (ALH). This, in turn, compromises the accurate classification of sperm motility patterns. A recent study sought to address this challenge using frequency-domain smoothing techniques and deep learning models, offering a novel approach to sperm motility analysis.

PAW: A More Robust Indicator of Lateral Displacement
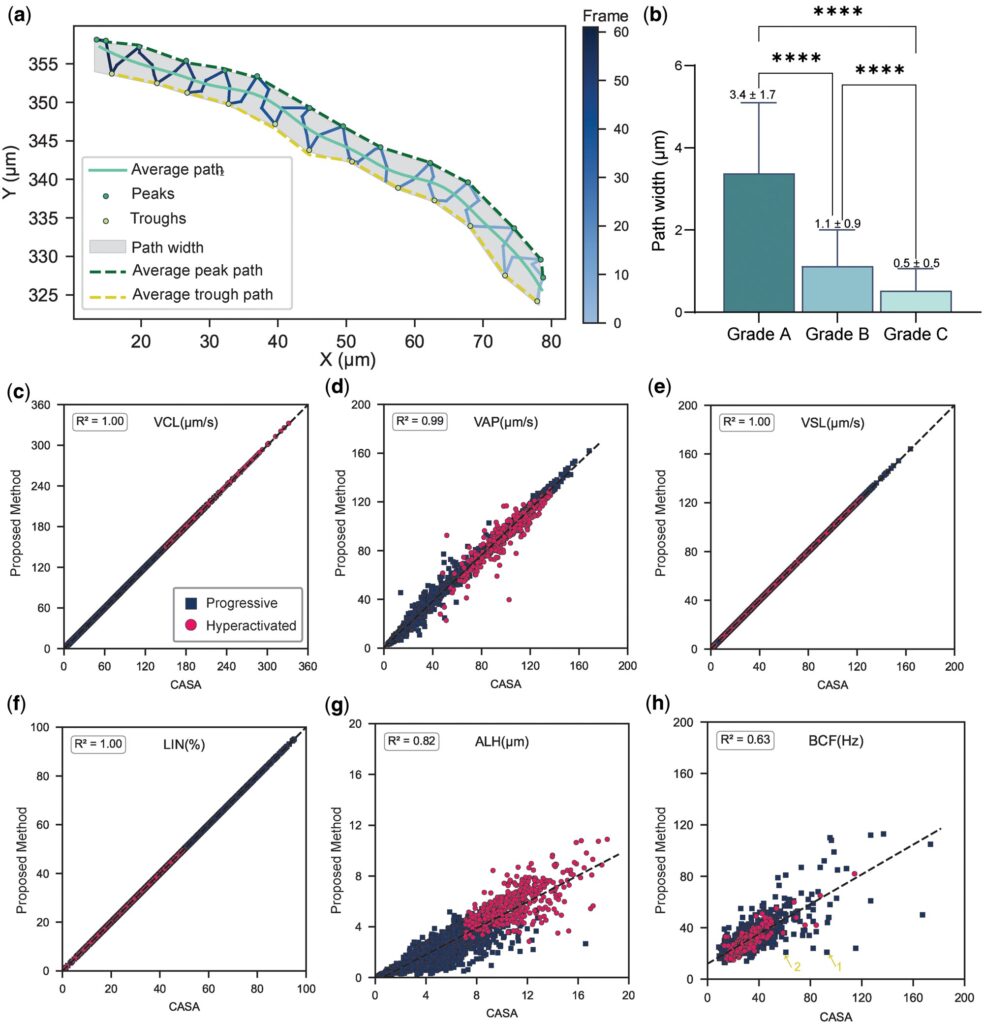
The research team focused on the limitations of current sperm motility assessment methods. Lateral displacement indicators reported by conventional systems only capture the maximum amplitude of sperm head oscillation, failing to comprehensively reflect actual sperm movement characteristics. The investigators proposed a new metric—Path Average Width (PAW)—to describe the overall lateral distribution range of a sperm’s trajectory relative to its mean path. Unlike existing indicators, PAW characterizes the lateral spread of the trajectory throughout the entire movement process, rather than merely recording the maximum oscillation amplitude.
The innovative significance of this indicator lies in its ability to capture high-frequency features of sperm flagellar beating, which are closely associated with complex motility patterns such as hyperactivation. During fertilization, hyperactivated sperm require vigorous flagellar movement to penetrate the zona pellucida of the oocyte, and elevated PAW values may reflect this specialized functional demand. Consequently, PAW is expected to serve as a more sensitive biomarker for assessing sperm fertilization potential.
Study Results
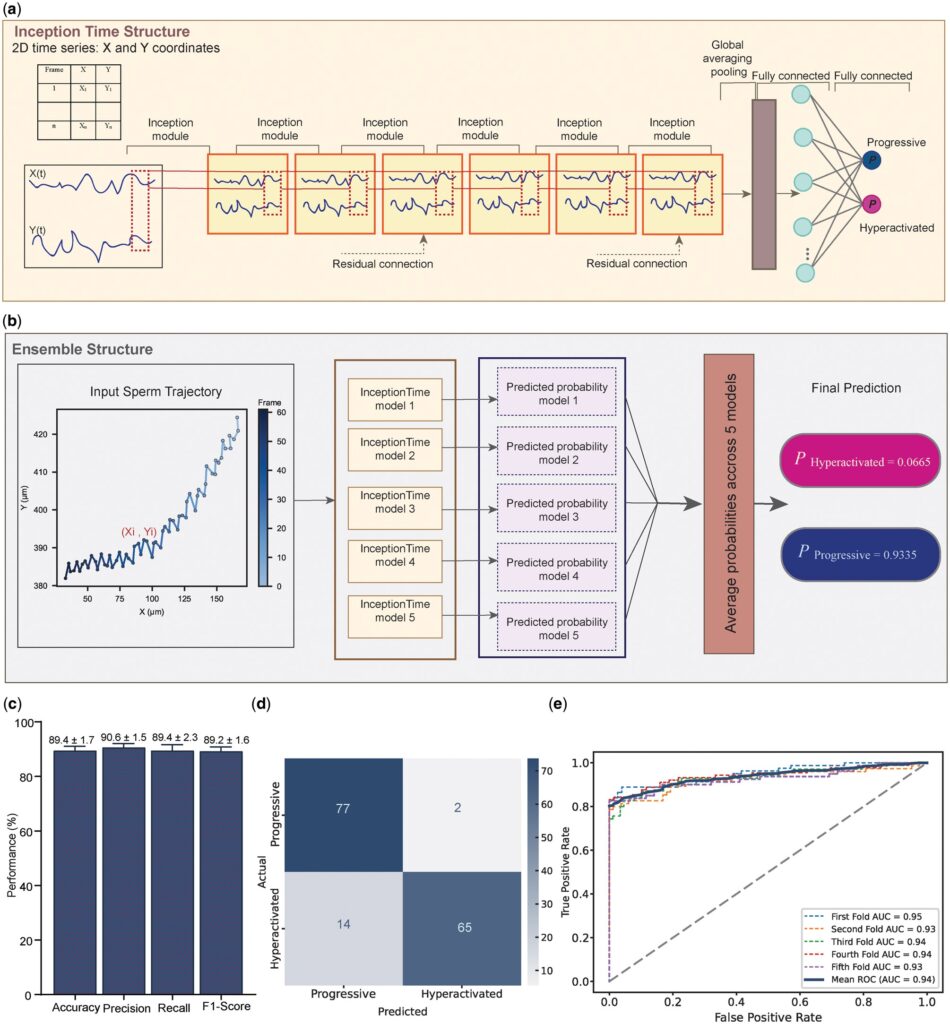
The study enrolled hundreds of semen samples, each containing thousands of sperm movement trajectories. Trajectory data were processed using the DCT-12 frequency-domain smoothing algorithm to eliminate interference caused by stroboscopic light sources. The deep learning model achieved 89% accuracy and an AUC of 0.94 for the binary classification of progressive and hyperactivated sperm. For the multi-class classification of WHO motility grades (Grades A, B, and C), the model attained 78% accuracy.
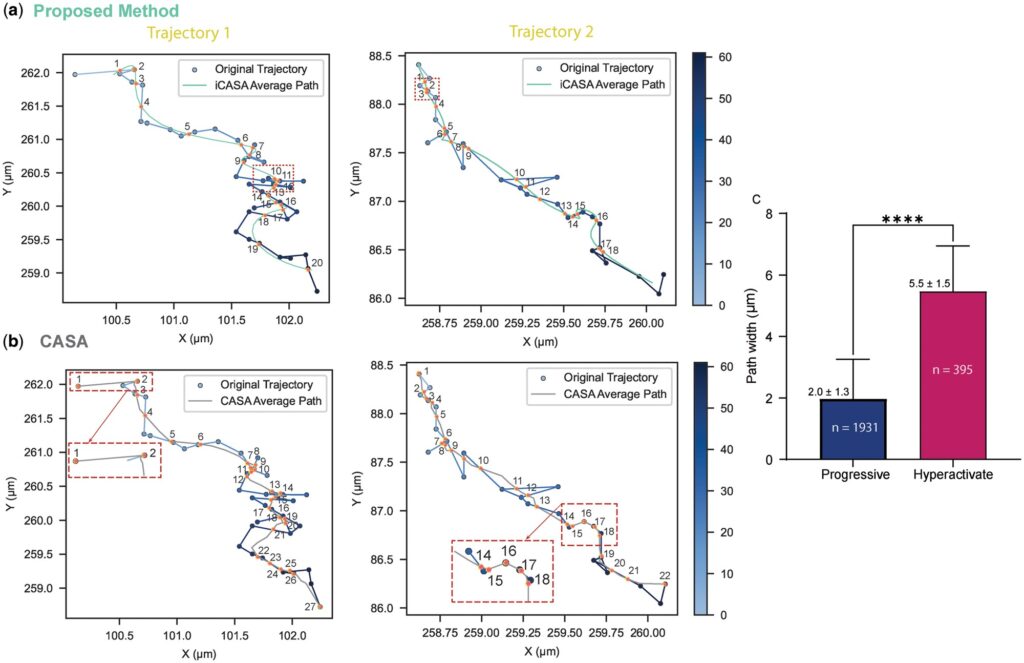
The PAW indicator performed prominently: hyperactivated sperm exhibited a PAW of 5.5±1.5 μm, significantly higher than the 2.0±1.3 μm observed in progressive sperm (P<0.0001), demonstrating its effective discriminatory power for sperm motility patterns. In the independent validation set, the model achieved a classification accuracy of 96%, indicating favorable generalizability.
Limitations and Future Perspectives
This study establishes a novel technical framework for sperm motility analysis, yet it has several limitations. First, although the dataset encompasses tens of thousands of trajectories, further expansion is required to train more stable and universally applicable deep learning models. Second, the correlation between PAW values and actual pregnancy outcomes remains unvalidated—whether higher PAW values necessarily translate to improved fertilization and pregnancy rates requires prospective clinical investigations. Additionally, the study was primarily conducted using conventional semen samples; its applicability in patients with oligoasthenozoospermia or surgically retrieved sperm specimens warrants further verification.
The introduction of the PAW indicator enriches the parameter system for sperm motility assessment and provides a new perspective for interpreting complex movement patterns. Deep learning demonstrates the capacity to automatically learn classification features from raw trajectory data, potentially reducing biases arising from manually defined rules. Further research is needed to validate the clinical value of this novel technology and facilitate the development and implementation of user-friendly clinical tools.
Conclusion
This study improves the processing precision of sperm movement trajectories using the DCT-12 frequency-domain smoothing algorithm and introduces the PAW metric to evaluate sperm lateral displacement characteristics. The integration of deep learning achieves 89% accuracy in classifying progressive and hyperactivated sperm, with PAW values showing significant differences between the two groups, indicating excellent discriminatory performance.
For embryologists, the clinical implications are twofold:
- The PAW indicator expands the parameter set for sperm motility evaluation, offering new insights into complex movement patterns and enabling a more comprehensive assessment of sperm quality.
- Deep learning automates feature extraction from raw trajectory data, minimizing subjective biases from manual rule-setting and supporting more objective clinical decision-making.
While further validation of the link between PAW and pregnancy outcomes is necessary, this research direction holds substantial promise and merits continued investigation.

